The production of aseptic pharmaceutical products has historically been the most demanding and risky pharmaceutical project. Many pharmaceutical companies are faced with the "dilemma" of changing the production process or abandoning the production of aseptic products that cannot be terminally sterilized.
This article will compare the traditional aseptic filling process, freeze drying process, and bfs aseptic filling process to tell you why ASEPTIC BFS MACHINE IS YOUR BEST CHOICE.
Aseptic BFS Filling Process:
The emergence of the Blow Fill Seal “three-in-one” aseptic filling process (hereinafter referred to as the BFS process) should be marked by the BFS machine invented by German engineer Gerhard Hansen in 1962. The BFS machine can complete the CIP/SIP (on-line cleaning/on-line sterilization) of all material pipelines under the control of computer programs, so that the three processes of bottle making, filling and sealing are completed in the same sterile environment under the protection of Class A air shower.
The entire production control process is completed by the computer program according to the predetermined program. The possible risks (environment, temperature, pressure difference, etc.) in the process are monitored by the computer control system according to the set parameters. The parameters are completely stored in the computer and can be consulted at any time, but cannot be changed. This is the filling process with the strongest sterility guarantee capability at present.
The BFS Aseptic Filling Technology Process is Shown in Figure 1.
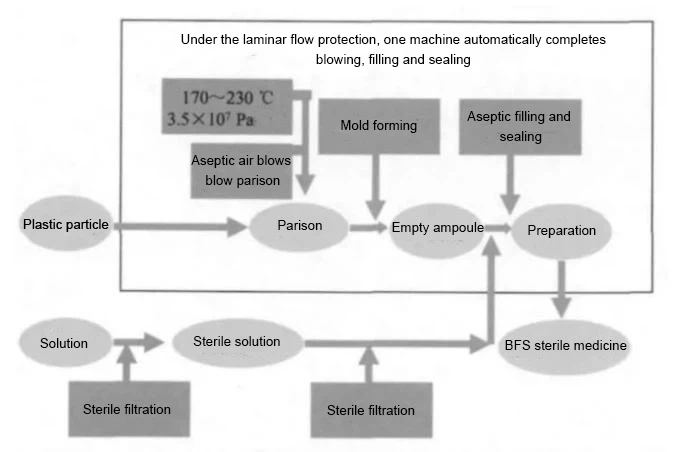
Introduction of the 8 detail advantages you need to know about aseptic BFS machine filling technology.
1. Superior aseptic production environment design:
 BFS machine is designed and installed according to the principle of “black and white partition”. The whole production process is controlled by the computer program, and the process parameters are stable and reliable. The filling area of the equipment is protected by A-level laminar flow, there is no operator in the filling room during production, and there is no pollution source in the production process. All process pipelines (including the preparation system) in contact with materials can realize CIP/SIP, and the blowing, filling and sealing stations are protected by A-level air showers, products are trimmed outside the filling room. One side of the product conveying channel is in the A-level laminar flow area and the other side is in the general production area, there is a pressure difference protection of not less than 40Pa, and the entire process has a strong aseptic guarantee capability.
BFS machine is designed and installed according to the principle of “black and white partition”. The whole production process is controlled by the computer program, and the process parameters are stable and reliable. The filling area of the equipment is protected by A-level laminar flow, there is no operator in the filling room during production, and there is no pollution source in the production process. All process pipelines (including the preparation system) in contact with materials can realize CIP/SIP, and the blowing, filling and sealing stations are protected by A-level air showers, products are trimmed outside the filling room. One side of the product conveying channel is in the A-level laminar flow area and the other side is in the general production area, there is a pressure difference protection of not less than 40Pa, and the entire process has a strong aseptic guarantee capability.
2. Super cost saving ability
The BFS machine makes the container in aseptic state without cleaning and sterilizing the container, saving process water and energy.
3. High product quality and comprehensive economic benefits
BFS machine adopts modular design, one equipment is like a small factory, which can automatically complete the whole process and monitoring of blowing, filling and sealing. The equipment occupies a small area and consumes less energy in the production process. Therefore, the comprehensive production cost of the BFS aseptic filling process is not higher than that of the traditional aseptic filling process, but the product quality and comprehensive economic benefits are much higher than the traditional aseptic filling process.
4. Ultra-high technical content and product added value
BFS machine can use PP/PE2 materials according to the process needs, the comprehensive cost of plastic container production is low, and the product technology content and added value are high.
5. Computer control, safe and reliable results
BFS machine can automatically complete CIP/SIP under computer program control, with reliable reproducibility and safe and reliable results.
6. Suitable for the production of various forms of aseptic filling products
BFS equipment can realize aseptic filling without high temperature sterilization, and the product quality is stable. It can be applied to various forms of aseptic product filling and the production of various aseptic containers only by changing different molds.
7. Aseptic production, aseptic use, cost savings
The filling machine of BFS equipment forms a "Luer interface" structure at the bottle neck while the product is being filled. The "Luer interface" can be tightly connected with the syringe. When the solution is extracted, the external air will not enter the container, and products can realize aseptic production, aseptic use, and good safety performance. Plastic packaging waste is easy to handle and will not pollute the environment.
BFS aseptic filling process has a history of nearly 50 years. Due to the advantages of strong sterility guarantee in the process of production and use, it has become a trend to replace the glass ampoule production process of washing, filling, sealing and sterilization and the glass bottle infusion production process with the BFS aseptic filling process. Flexible packaging aseptic products have irreplaceable advantages in special fields such as combat readiness and disaster relief.
8. The BFS process is more suitable for the production of heat-sensitive drugs, biochemical preparations, vaccines and other products.
During the production process, the unique mold design makes the forming and cooling process of the ampoules complete almost simultaneously. Although the temperature of the product rises slightly at the beginning of filling, it is not enough to affect the quality of the drug.
Traditional Aseptic Filling Process:
In the traditional aseptic filling process, since the equipment cannot truly realize CIP/SIP, especially the key components of the equipment must be manually assembled and debugged before use, which will cause pollution to the equipment and the aseptic environment. Containers and components are purchased from outside, and they need to be cleaned and sterilized separately, and then combined together. There is a risk of contamination in each link, and it is impossible to meet the requirements of sterility assurance throughout the process.
The actual production process also confirmed the defects of traditional aseptic filling. For example, the washing, filling, sealing and sterilization processes of small-volume glass ampoules have the following defects:
1. High comprehensive cost:
(1) For the outsourced glass ampoules, it is necessary to build a larger warehouse for inner packaging materials.
(2) There are many washing, filling, sealing, sterilizing and post-processing equipments, the production line is long, the workshop area is large, and the infrastructure cost is high.
(3) The clean production area is large, the area division is complex, and the control and detection are difficult.
(4) The bottle washing process requires a lot of water, and the containers and finished products need to be sterilized twice, which consumes a lot of energy.
(5) There are many operating positions, and the management risk is high.
(6) The product is easily broken, and the packaging and transportation costs are high.
The comprehensive cost of traditional aseptic filling process is higher than that of BFS aseptic filling process.
2. Contamination risk:
(1) Flame hot-melt sealing, negative pressure will be generated in the ampoule when it is cooled, and a large amount of fine glass chips will enter the liquid under the action of negative pressure, which will contaminate the product during use, and insoluble particles will cause potential damage to users. There are risks in the use of the product.
(2) Sharp glass ampoule breaks can also cause injury to the operator.
3. Risks are also existed in high temperature sterilization:
(1) The high-temperature sterilization process will change the active ingredients of some medicines, and at the same time generate "new substances", causing phytotoxic events.
(2) Some products cannot be sterilized at high temperature due to the limitation of raw materials and process, and do not meet the requirements of sterile drug production process.
(3) Circulation steam sterilization" itself is a very risky process.
4. Other disadvantages
(1) Equipment and process pipelines, especially the equipment components and conveying pipelines after sterilization and filtration, cannot achieve CIP/SIP.
(2) The components of the equipment before filling need to be manually assembled and debugged, and the filling area needs to be operated by personnel. There are many uncontrollable factors, and the aseptic guarantee ability is not good.
(3) Non-degradable waste will pollute the environment and fail to meet the requirements of user safety, operator safety and environmental safety.
Freeze Drying Process
Many products cannot achieve terminal sterilization due to heat resistance, so they have to choose the freeze-drying process. BFS aseptic filling process and freeze-drying process are both non-terminal sterilization processes. By comparing the aseptic guarantee capability, production efficiency and production benefit of the production process, it can be seen that the BFS aseptic filling process has great product safety and incomparable technological advantages compared with the freeze-drying process.
The basic process of freeze-drying technology: vial bottles and rubbers cleaning and sterilizing → product filling → semi-stoppering → long-term freezing sublimation in freeze dryer, press stopper → out of freeze dryer → capping aluminum cap → labeling and packaging.
The production of freeze-dried products requires the purchase of vials, rubber stoppers, and aluminum caps, and requires a large area of warehouse for storage. The freeze-drying process has many equipment, high-grade clean workshops occupy a large area, high requirements, difficult control, and large investment. The freeze-drying process route is long, the production process cannot be unmanned, the product cannot be effectively leaked, and insoluble particles cannot be detected. There are many links and high risks; long production cycle, high cost, low output and economic benefits.

